The Medical Device Manufacturing Toolkit
Your resource for navigating the complex
world of regulations, compliance, and total
security across the medical device lifecycle.
DIGICERT IoT TRUST MANAGER
Case Study: B. Braun
Patients in more than 160 countries count on connected medical devices built by B. Braun. And B. Braun counts on DigiCert for digital trust.
Learn More

CHECKLIST
The 7 Habits of Trustworthy Medical Devices

SOLUTION BRIEF
Digital Trust in Connected Healthcare

EBOOK
Device Trust: Securing the Future of Smart Technology

BLOG
Healthcare Data and Quantum Computing

BLOG
Securing Diabetes Monitors

BLOG
Securing Next Gen 9-1-1

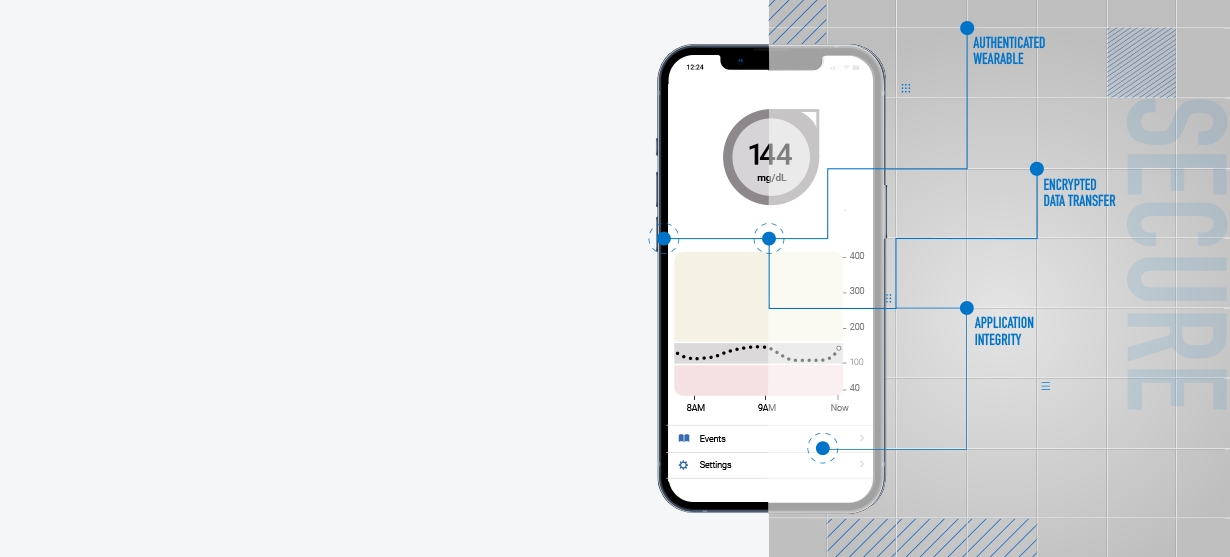
Connected Healthcare Threats can Have Catastrophic Consequences
The rise in in-home medical device use offers patients more medical flexibility than ever before. But more connections mean more attack vectors, posing a massive threat to patients and the manufacturers and providers the public entrusts with their lives.
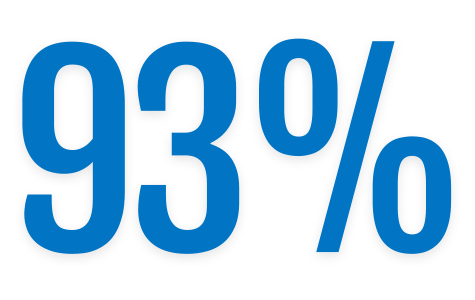
2024 State of Digital Trust Report
of the device manufacturers surveyed have experienced
- Data breaches
- Outages
- Brownouts
